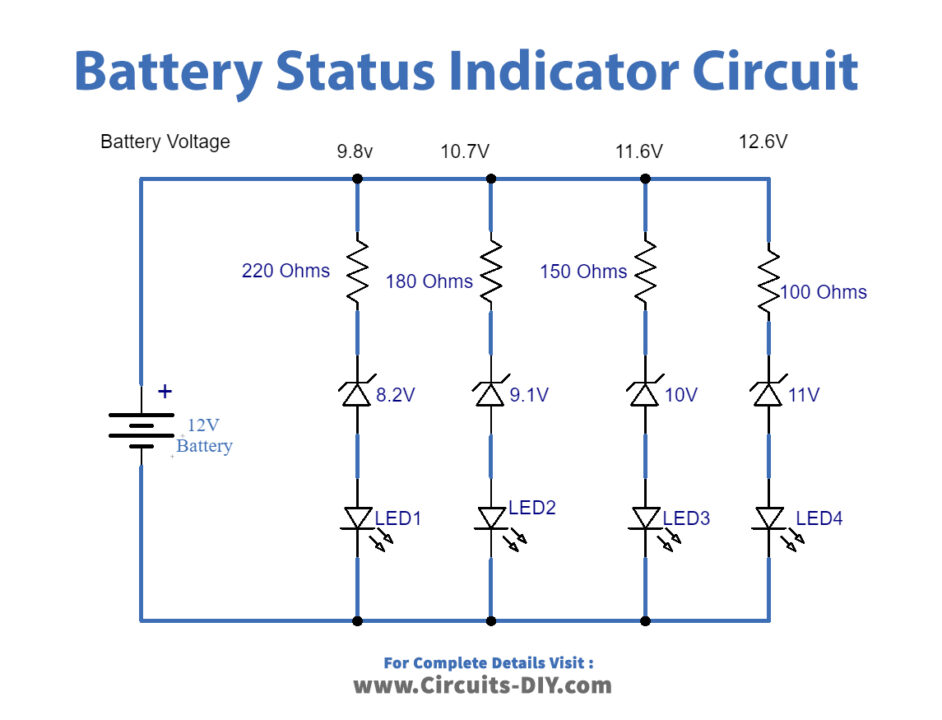
Thus, internal resistance reduces both the current and voltage available to the load. When current flows in the circuit (I L ), the internal voltage drop (I LR i) drops the terminal voltage of the battery as shown in below Equation. If a load is placed on the battery, load resistance (R L) is in series with internal resistance (R i).įigure 9 : Internal Resistance in a Chemical Cell With no current flow, the voltage drop is zero thus, the full battery voltage is developed across the output terminals (V B). The internal resistance is in series with the voltage of the battery, causing an internal voltage drop (Figure 10). Internal resistance in a chemical cell is due mainly to the resistance of the electrolyte between electrodes (Figure 9).Īny current in the battery must flow through the internal resistance. 
For example, a 120 ampere-hour battery must be recharged after 12 hours if the discharge rate is 10 amps. The capacity of a storage battery determines how long the storage battery will operate at a certain discharge rate and is rated in ampere-hours. The most common example of a secondary, or rechargeable cell, is the lead-acid automobile battery. Secondary CellsĬells that can be recharged to nearly their original condition are called secondary cells. Dry cells that are used in flashlights and transistor radios (e.g., AA cells, C cells) are examples of primary cells. The advantage of connecting cells in parallel is that it will increase the current-carrying capability of the battery.įigure 8 : Cells Connected in Parallel Primary CellĬells that cannot be returned to good condition, or recharged after their voltage output has dropped to a value that is not usable, are called primary cells. The total voltage output of a battery connected in parallel is the same as that of a single cell.Ĭells connected in parallel have the same effect as increasing the size of the electrodes and electrolyte in a single cell. When cells are connected in parallel, all the positive terminals are connected together, and all the negative terminals are connected together. The current flow through a battery connected in series is the same as for one cell.įigure 7 : Cells Connected in Series Parallel CellsĬells connected in parallel (Figure 8), give the battery a greater current capacity. When we connect cells in series, the positive terminal of one cell is connected to the negative terminal of the next cell. In the example of the battery in Figure 7, the four 1.5V cells provide a total of 6 volts. 
When several cells are connected in series (Figure 7), the total voltage output of the battery is equal to the sum of the individual cell voltages. Once the basic theory behind the operation of batteries is understood, we can apply these concepts to better understand the way batteries are utilized.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
